The Technology of the Future.
The plasma source, which forms the basis of our technology, generates plasma as the fourth state of matter—alongside solid, liquid, and gas. It requires electrical energy and ambient air to create the plasma state and cold plasma products within this gaseous environment. Cold plasma products are made from electrically charged particles. They are primarily reactive oxygen compounds with oxidation potential, which eliminate harmful microorganisms. Pathogenic organisms such as bacteria, viruses and fungi are reliably oxidized. Our PLASMO®AIR and PLASMO®CAR products are based on this technology.
As part of our research, cold plasma technology has been further developed into Cold Plasma and Aerosol for specific areas of application. In the scientific literature, this approach has been termed Cold Atmospheric Plasma-Aerosol (CAP-A). Cold plasma products interact with pre-nebulized water: the aerosol. The degree of ionization of the cold plasma enhances the effectiveness of the nebulized water. It works according to the following process: Reactive Oxygen Species (ROS) generated as part of cold plasma electrostatically charge the aerosol and, through this dispersion and their intrinsic properties, increase the antimicrobial efficacy of Cold Atmospheric Plasma-Aerosol (CAP-A). This way, the patented process especially amplifies the physical dissolution and displacement effect of the aerosol. Our products PLASMO®HEAL PRO, PLASMO®VET, and PLASMO®HAND benefit from this advancement.

Mode of Action of Cold Atmospheric Plasma (CAP).
Cold Atmospheric Plasma (CAP) consists in an ionized gas. In addition to solid, liquid and gaseous, this “electrically charged” gas represents a fourth state of matter as outlined above. Cold plasma products contained in this gas consist of various charged particles with oxidation potential. They enable the oxidation of unwanted microorganisms. The charged particles move freely in space and have their desired effect there. Cold Atmosperhic Plasma (CAP) is also required to produce Cold Atmospheric Plasma-Aerosol (CAP-A). The exact explanation of how CAP is produced can therefore be found in the next section “Step 1. Generating Cold Atmospheric Plasma (CAP)”.
Mode of Action of Cold Atmospheric Plasma-Aerosol (CAP-A).

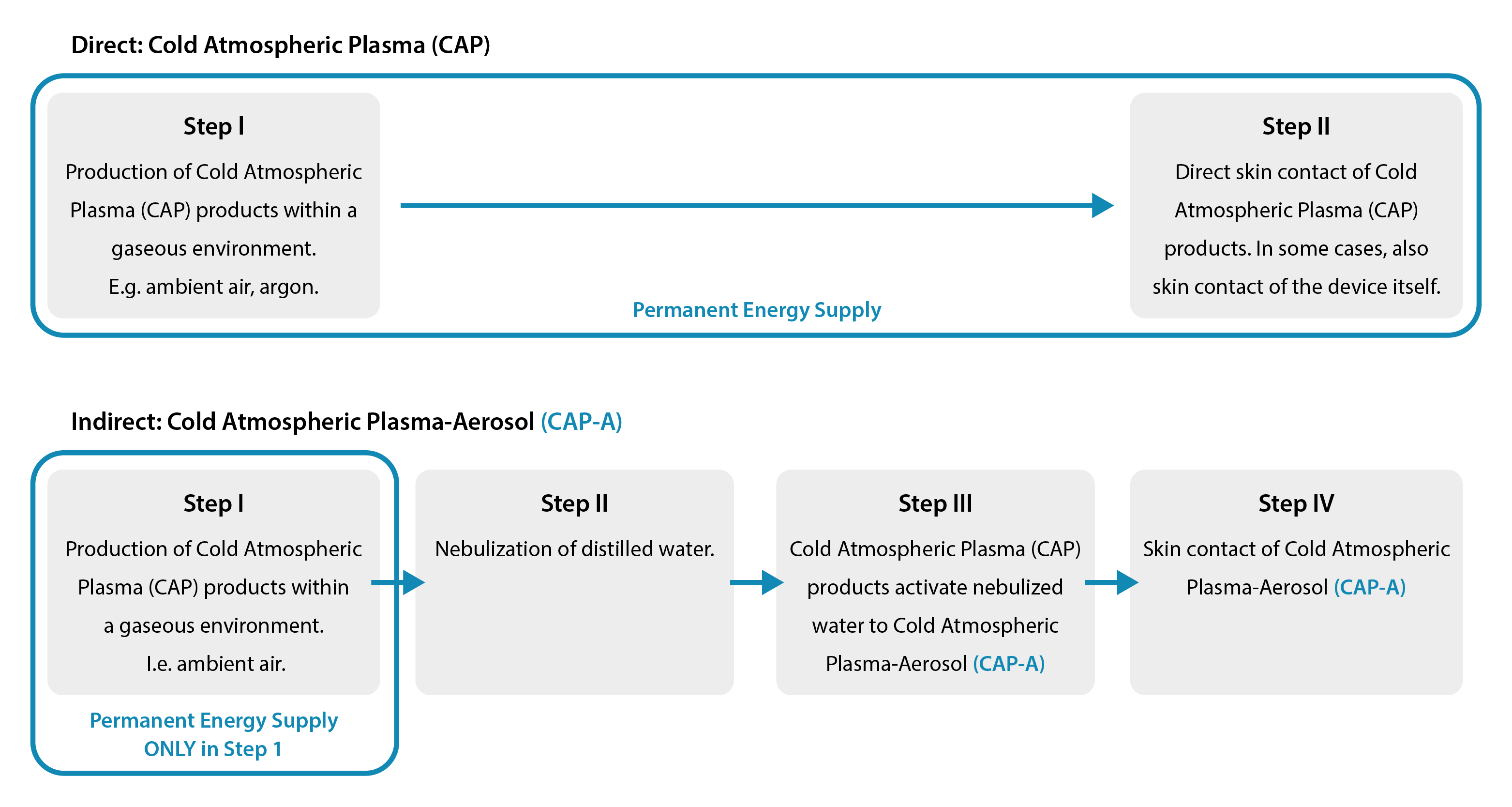
Our products PLASMO®HEAL PRO, PLASMO®VET and PLASMO®HAND are based on the further development of the direct cold plasma process for medical applications. The indirect cold plasma process is referred to as Cold Atmospheric Plasma Aerosol (CAP-A) in scientific literature. In addition to cold plasma products, CAP-A uses the power of the aerosol. This process differs from the direct Cold Atmospheric Plasma (CAP) process because it does not require a permanent input of energy. Energy supply and area of application are separated from each other. The following steps first explain the generation of Cold Atmospheric Plasma (CAP) and, building on this, the generation of Cold Atmospheric Plasma-Aerosol (CAP-A).
Step 1.
Generating Cold Atmospheric Plasma (CAP).
Plasma Disinfection Procedures for Surfaces in Emergency Service Vehicles: A Field Trial at the German Red Cross. Sci Rep 13, 20737.
DOI: https://doi.org/10.1038/s41598-023-47759-5
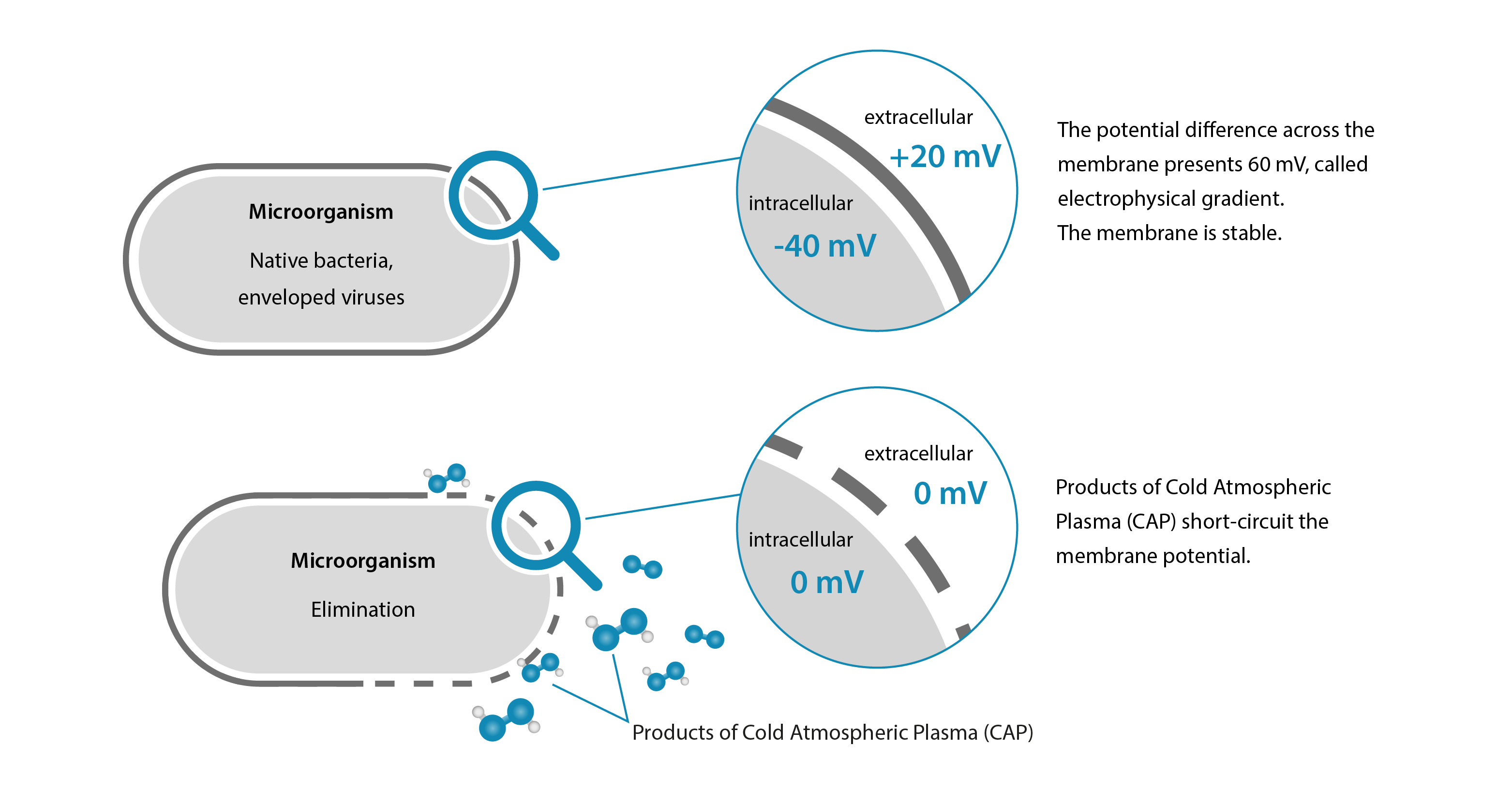
The core technology presents Cold Atmospheric Plasma CAP). Its generation is based on a plasma source in each device. The plasma source requires only electricity and ambient air. This is how cold plasma is produced — an ionized gas and fourth state of matter. The within the gaseous phase contained cold plasma products consist of various charged particles with oxidation potential. These enable the oxidation of microorganisms such as native bacteria, viruses, fungi and multi-resistant germs (MRSA). Cold plasma products are Reactive Oxygen Species (ROS) which cause a short circuit of the cell membrane of the pathogenic microorganism. Specifically, these particles damage the cell membrane. Redox processes of microorganisms are then disrupted. Pathogenic microorganisms are thus inactivated. Human and animal cells are protected by proteins in their cell network and are therefore not affected — hence, Cold Atmospheric Plasma (CAP) is harmless to humans and animals. This germ-inactivating effect also supports the automatic decontamination of the device. In addition to the inactivation effect, we make particular use of the ionization degree of Cold Atmospheric Plasma (CAP), which becomes relevant in the third step.
Step 2.
Aerosol Production.
In PLASMO®HEAL PRO/ PLASMO®VET/ PLASMO®HAND, a nebulizer module produces a very fine aerosol in the second step. Our aerosol consists of smallest water particles and ambient air.
The cold plasma products produced in step 1 hit the aerosol generated in step 2 in a chamber. The oxygen compounds of the cold plasma products electrostatically charge the aerosol. This is made possible by the degree of ionization of the plasma. The patented mixture of cold plasma products and nebulized water (aerosol) promises high effectiveness. The electrostatic charging of the droplets ensures that the aerosols are evenly distributed. The power of the aerosol dispersion is increased.
In contact with the skin, the conditioned aerosol develops the power of its dissolution and displacement effect with regard to microorganisms in this fourth and final step. This main physical mode of action results in mechanical decontamination of the skin. The graphic below shows how treatment with Cold Atmospheric Plasma (CAP-A) effectively inactivates transient bacteria on the surface. The desired bacteria of the body's own microbiome — the resident flora — are not affected. In addition, osmolarity, pH value reduction and Reactive Oxygen Species (ROS) have a favorable effect.
Cold Atmospheric Plasma (CAP), produced in a first step, comes into contact with the skin when energy is constantly supplied (e.g. “beam”) regarding dermatologic application. In contrast, Cold Atmospheric Plasma-Aeorosl (CAP-A) separates energy input and application physically and spatially. Energy is only required in the first step to produce cold plasma products at the plasma source. The targeted application of Cold Atmospheric Plasma-Aerosol (CAP-A) to the skin in the fourth and final step is completely separated. Our process is different from the direct cold plasma process because it does not involve any energy input into the skin. Energy supply and skin are separate from each other. It is precisely this separation that guarantees pain-free and stress-free care.
This technology offers decisive advantages in terms of effectiveness and application — both in medicine and in industry.
Plasma Disinfection Procedures for Surfaces in Emergency Service Vehicles: A Field Trial at the German Red Cross. Sci Rep 13, 20737.
DOI: https://doi.org/10.1038/s41598-023-47759-5